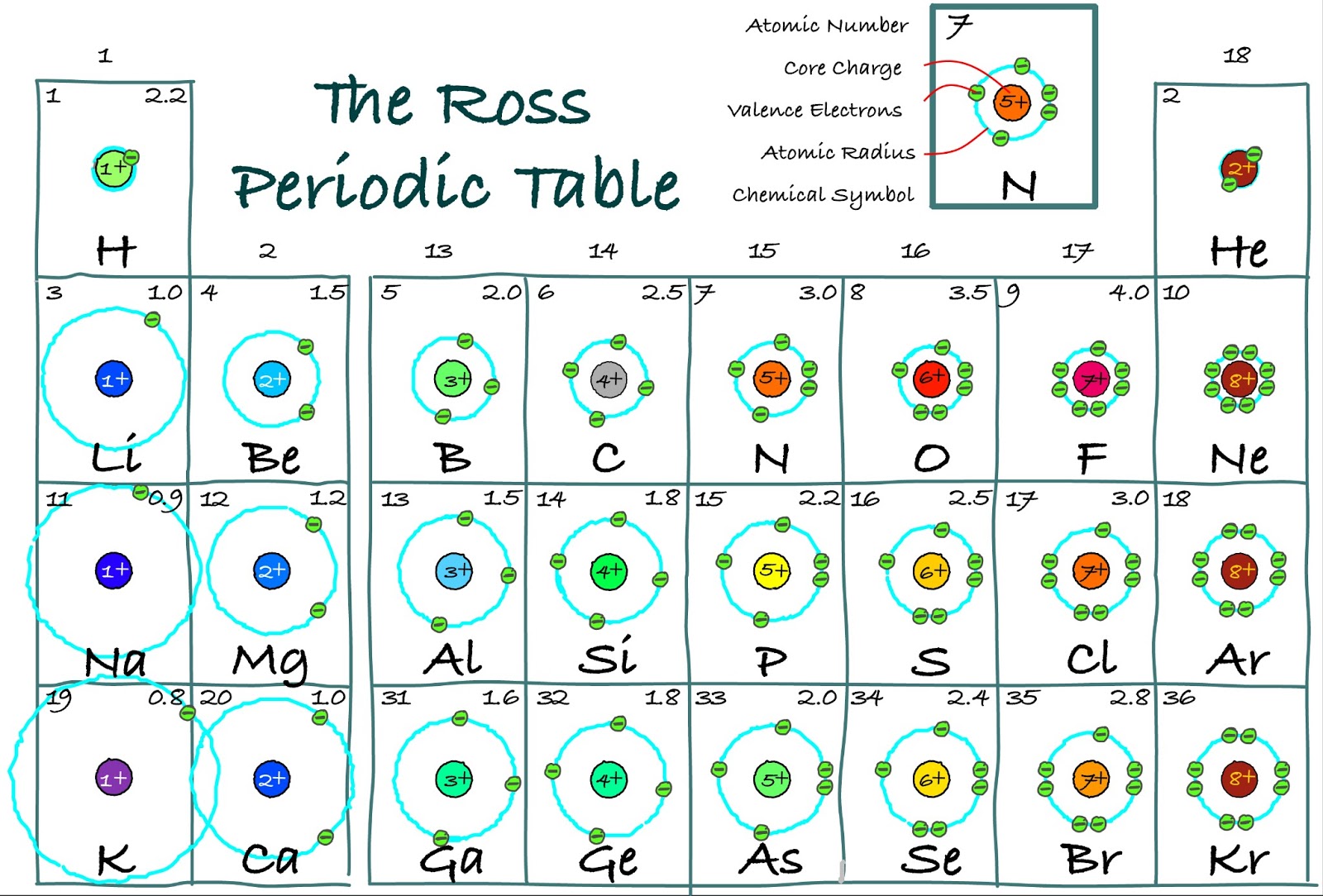
The same exact logic goes to find that Gallium is in the fourth period and the thirteenth group. So silicon is in the third row and fourteenth column, or third period and fourteenth group. This periodic table has the electron configuration for each row written along the left. Because they are in the outer shells of an atom, valence electrons play the most important role in chemical reactions. It's not shown in the linked table, but the group that this element is in is shown at the top of the table it's also just the number of the column counting from left to right. When their electron configurations are added to the table (Figure 6), we also see a periodic recurrence of similar electron configurations in the outer shells of these elements. The element in the second column of the p-orbitals and third row would be silicon (Si). Because there are two electrons in the p-orbital, the column (or group) that this element is in will be the second in the 3p row. Electron configurations can be determined using a periodic. Draw an orbital diagram and use it to derive the electron configuration of phosphorus, Z 15. The arrangement of electrons in the atomic orbitals of an atom is called the electron configuration. By knowing the electron configuration of an element, we can predict and explain a great deal of its chemistry. The table linked below shows which row (or period) on the table has elements with valence 3p orbitals. The electron configuration of an element is the arrangement of its electrons in its atomic orbitals. An element with the valence shell configuration 3s 23p 2 has a full s-orbital (because s-orbitals can hold at most two electrons) and two electrons in the p-orbitals. 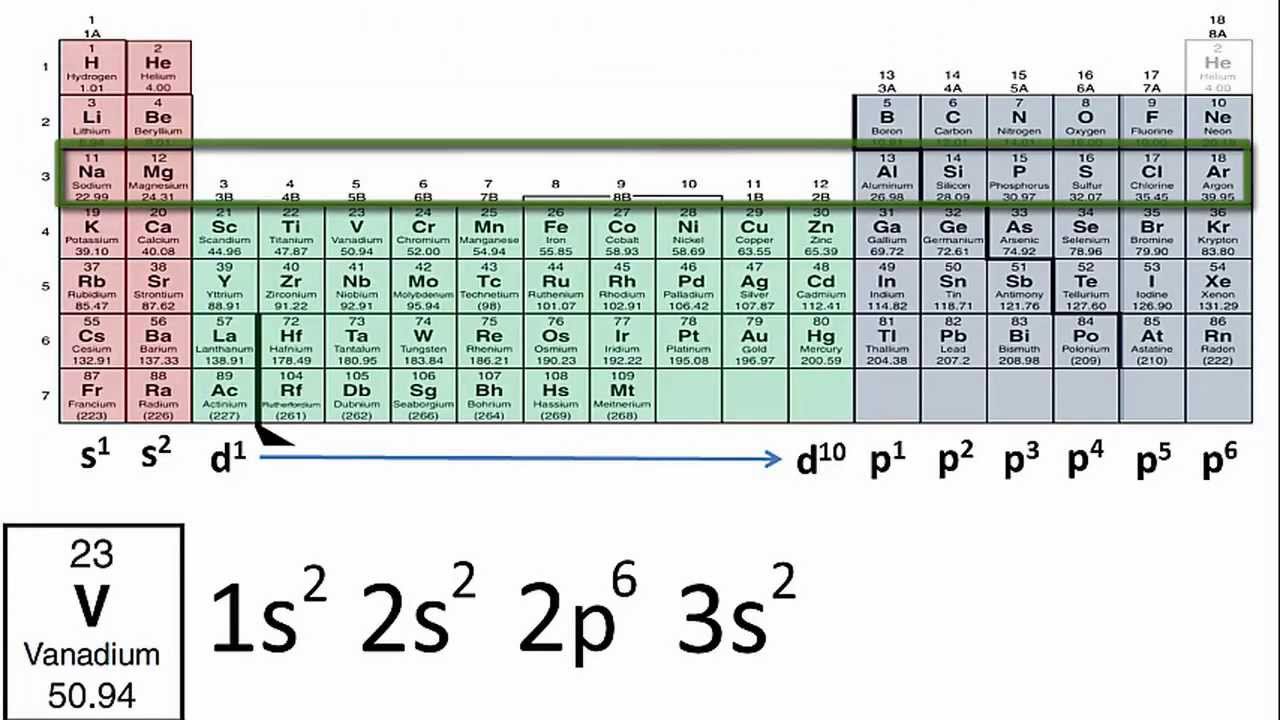
If you take a look at a periodic table that shows orbitals 1 you can see what elements have which configurations. Valence electrons of Elements (List) Here is the list of periodic table of elements with their valence electrons. Excluding the transition metals and helium, any element on the periodic table will have the same number of valence electrons as the final digit of the group.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
